43 keppra off label
Use of Anti-Epileptic Drugs for Off-Label Indications and the increasing evidence of off-label use of ... o Three drugs, levetiracetam (Keppra), felbamate (Felbatol), and tiagabine (Gabitril) had most of their ... Anti-Epileptic Drugs and Medications - Verywell Health Oxcarbazepine is sold under the brands Trileptal, Oxtellar, and Oxtellar XR. It is used for adults, and for children ages 2 and older, to treat partial epilepsy. It can be used alone or in combination with other medications. 11. Oxcarbazepine is available as a tablet, an extended-release formula, and an oral liquid.
(PDF) Off-Label Drug Uses - Levetiracetam: Rapid-Cycling Bipolar ... Off-Label Drug Uses - Levetiracetam: Rapid-Cycling Bipolar Disorder (Adults). May 2009; Hospital Pharmacy 44(5):390-391. DOI:10.1310/hpj4405-390.
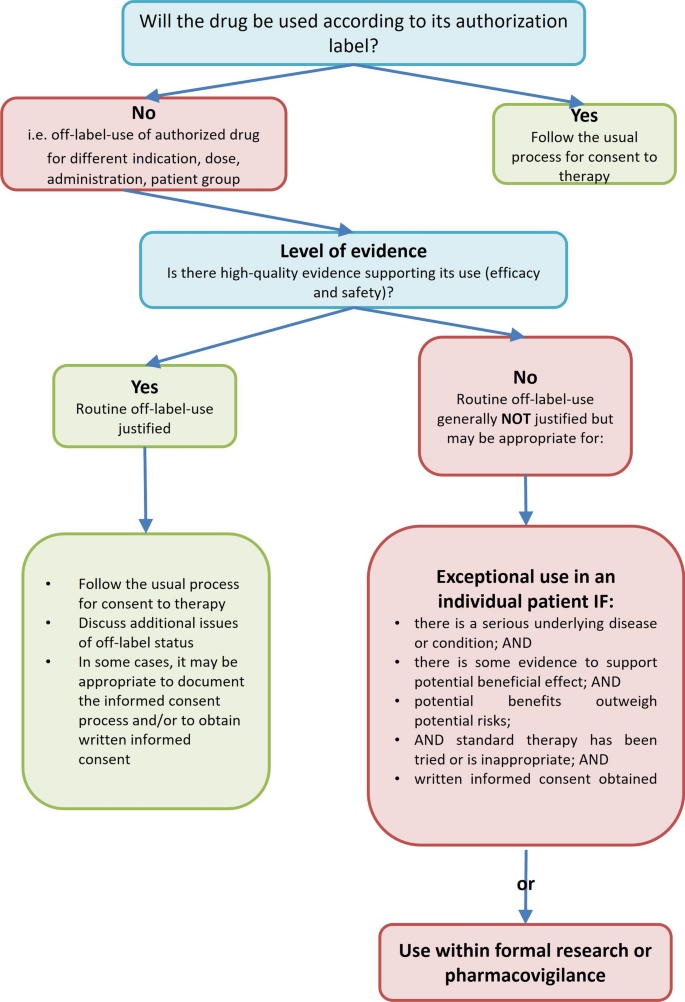
Keppra off label
Off-label use of antiepileptic drugs for the treatment of neonatal ... Seventy-three percent (40/55) recommended treatment of neonatal seizures with one or both of levetiracetam and topiramate; 47% (26/55) recommended levetiracetam ... LevETIRAcetam: Dosage, Mechanism/Onset of Action, Half-Life - Medicine.com Keppra: 100 mg/mL (473 mL) [gluten free, lactose free; contains methylparaben, propylparaben; grape flavor] Generic: 100 mg/mL (5 mL, 473 mL, 500 mL [DSC]) Tablet, Oral: Keppra: 250 mg [scored; contains fd&c blue #2 aluminum lake] Keppra: 500 mg [scored] Keppra: 750 mg [scored; contains fd&c yellow #6 aluminum lake] Keppra: 1000 mg [scored] Wary off-label Keppra patient | Epilepsy Foundation off label use is always a concern. do some research and see what you think. if it control the bi-polar symptom, there will be more to be considered. hope it helps. rikk Re: Wary off-label Keppra patient Submitted by NotafanofBran on Thu, 2008-12-11 - 03:22
Keppra off label. Monotherapy treatment of bipolar disorder with levetiracetam When polypharmacy is unsuccessful, the clinician must consider the off-label use of newer psychotropics. Levetiracetam is a novel anticonvulsant with antikindling, inhibitory, and neuroprotective properties that is effective in an animal model of mania. keppra off label use - MedHelp This medication is typically used to treat seizure disorders, but does have off label uses such as neuropathic pain. Abrupt discontinuance of the drug may cause seizures. You said that you are not taking any anti-seizure medications right now, so I take that to mean you are not taking the Keppra right now? Keppra Oral: Uses, Side Effects, Interactions, Pictures ... - WebMD Keppra 500 mg tablet. Color: yellow Shape: oblong Imprint: ucb 500 . This medicine is a white, oblong, scored, film-coated, tablet imprinted with "ucb 1000". Keppra 100 mg/mL oral solution. Levetiracetam | VCA Animal Hospitals Levetiracetam (brand names: Keppra®, Elepsia®, Spritam®) is an anticonvulsant used to treat seizures and epilepsy. In dogs, it is typically used in combination with other anticonvulsants, while in cats, it is used alone or in combination. Its use in cats, dogs, and horses to treat seizures and epilepsy is 'off label' or 'extra label'.
Keppra Critical care Use - Neuroland 2G Using Keppra in special situations. All off label; ref: Uptodate; Craniotomy, seizure prophylaxis. 500 mg bid iv, up to 3 gram per day; Status epilepticus. Loading dose. 1 to 3 gram at 5 mg/kg/minute or; 50 mg/kg over 15 minutes, up to 4.5 gram; followed by maintenance dose; Subarachnoid Hemorrhage, Traumatic brain injury: short term seizure ... Keppra For Dogs: Side effects and Dosage - Relievet Key Takeaway: Keppra is an anti-seizure drug for dogs, cats, and people. It is often used in conjunction with Phenobarbital or Potassium Bromide. Its side effects include decreased appetite, drooling, vomiting, and behavioral changes. Its safety profile is considered better than Gabapentin's. With more than 5% of dogs suffering from seizures at ... Levetiracetam for Managing Neurologic and Psychiatric Disorders - Medscape It has since been approved for a wider patient population and is currently being used in the treatment of multiple seizure disorders, including generalized tonic-clonic; absence; myoclonic,... Levetiracetam - Wikipedia Levetiracetam, sold under the brand name Keppra among others, is a medication used to treat epilepsy. [6] It is used for partial-onset, myoclonic, or tonic-clonic seizures and is taken either by mouth as an immediate or extended release formulation or by injection into a vein. [6]
Keppra: Uses, Taking, Side Effects, Warnings - Medicine.com problems with muscle coordination (problems walking and moving) The most common side effects seen in people who take Keppra include: sleepiness. infection. weakness. dizziness. The most common side effects seen in children who take Keppra, in addition to those listed above, include: tiredness. decreased appetite. Pediatric Focused Safety Review: Keppra (Levetiracetam) - Food and Drug ... Keppra®(Levetiracetam XR tablets) • Adult approval: September 12, 2008 • Pediatric labeling: August 1, 2014* • ... • Off label use; drug administered to patient of FBI — U.S. Subsidiary of Belgian Pharmaceutical Manufacturer Pleads ... Keppra is not approved for the treatment of migraine, headache, psychiatric conditions or pain conditions. Once approved by the FDA, a manufacturer may not market or promote a drug for any use... PDF Reference ID: 4087271 - Food and Drug Administration KEPPRA is given orally with or without food. The KEPPRA dosing regimen depends on the indication, age group, dosage form (tablets or oral solution), and renal function. Prescribe the oral solution for pediatric patients with body weight 20 kg. Prescribe the oral solution or tablets for ≤ pediatric patients with body weight above 20 kg.
Keppra: Package Insert / Prescribing Information - Drugs.com Keppra is indicated as adjunctive therapy for the treatment of primary generalized tonic-clonic seizures in patients 6 years of age and older with idiopathic generalized epilepsy. Related/similar drugs gabapentin, clonazepam, lamotrigine, pregabalin, diazepam, Lyrica, topiramate Keppra Dosage and Administration Important Administration Instructions
National Center for Biotechnology Information Jun 24, 2022 ... Levetiracetam is a novel antiepileptic drug used to treat partial, myoclonic, and tonic-clonic seizures. In 2000, the FDA approved the use of ...
Levetiracetam (Keppra): Basics, Side Effects & Reviews - GoodRx Levetiracetam (Keppra) is an antiepileptic (anti-seizure) medication. It's not entirely clear how this medication works, but it helps calm certain nerves that are linked to seizures. What is levetiracetam (Keppra) used for? Partial-onset seizures
Keppra (Levetiracetam): Uses, Dosage, Side Effects ... - RxList KEPPRA tablets contain the labeled amount of levetiracetam. Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, polyethylene glycol 3350, polyethylene glycol 6000, polyvinyl alcohol, talc, titanium dioxide, and additional agents listed below: 250 mg tablets: FD&C Blue #2/indigo carmine aluminum lake
Keppra - GoToPills.com Keppra OFF-LABEL SEARCH Generic Name: Levetiracetam Drug Category: Antiepileptic Litigation Alert Level: High This drug has been approved for use by males and females over the age of 1 month old for a maximum duration of 3 years. Approved Uses Indicated for: Partial-Onset Seizures:
Keppra | European Medicines Agency Keppra is indicated as adjunctive therapy: in the treatment of partial-onset seizures with or without secondary generalisation in adults, children and infants from one month of age with epilepsy; in the treatment of myoclonic seizures in adults and adolescents from 12 years of age with juvenile myoclonic epilepsy;
View of Keppra a magical antiepileptic medication Keppra has been used as a broad spectrum anti-epileptic drug (AED). It is FDA approved for use in various types of partial and generalized seizures but its use ...
U.S. Subsidiary of Belgian Pharmaceutical Manufacturer Pleads Guilty to ... WASHINGTON - The U.S. subsidiary of Belgian pharmaceutical manufacturer UCB SA. pleaded guilty today to the off-label promotion of its epilepsy drug Keppra and will pay more than $34 million to resolve criminal and civil liability arising out of its illegal conduct, the Justice Department announced today.
Off-label prescribing of antiepileptic drugs in ... - PubMed Off-label prescribing of AEDs is common among patients with pharmacoresistant epilepsy and is influenced by demographic and disease-related characteristics. Studies are needed to improve the quality of evidence guiding epilepsy treatment, and to evaluate the risks and benefits of off-label prescribi …
All About Levetiracetam (Keppra) | Emergency Physicians Monthly Levetiracetam, brand named Keppra, is FDA-approved as adjunctive therapy to treat partial onset seizures in patients over one month of age, for tonic-clonic seizures in patients' age six years and up, and for myoclonic seizures in patients over 12 years old.
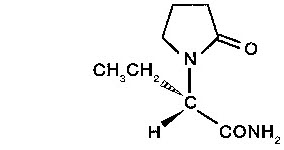
PDF KEPPRA (levetiracetam) Rx only 250 mg, 500 mg, 750 mg, and 1000 mg ... Levetiracetam is a white to off-white crystalline powder with a faint odor and a bitter taste. It is very soluble in water (104.0 g/100 mL). It is freely soluble in chloroform (65.3 g/100 mL) and in methanol (53.6 g/100 mL), soluble in ethanol (16.5 g/100 mL), sparingly soluble in acetonitrile (5.7 g/100 mL) and practically insoluble in n-hexane.
List of off-label promotion pharmaceutical settlements UCB: Off-label promotion of Keppra, June 2011. The U.S. subsidiary of UCB, a Belgian pharmaceutical company, agreed to pay more than $34 million as part of a settlement agreement to resolve all criminal and civil liability arising from its alleged off-label promotion of the epilepsy drug Keppra. UCB was alleged to have promoted the drug for the ...
Keppra Uses, Dosage & Side Effects - Drugs.com Keppra is a prescription medicine used to treat certain types of seizures in people with epilepsy, including partial onset seizures, myoclonic seizures, and tonic-clonic seizures. When used for partial onset seizures: Keppra XR is for adults and children at least 12 years old.
keppra off label uses - MedHelp This medication is typically used to treat seizure disorders, but does have off label uses such as neuropathic pain. Abrupt discontinuance of the drug may cause seizures. You said that you are not taking any anti-seizure medications right now, so I take that to mean you are not taking the Keppra right now?
Wary off-label Keppra patient | Epilepsy Foundation off label use is always a concern. do some research and see what you think. if it control the bi-polar symptom, there will be more to be considered. hope it helps. rikk Re: Wary off-label Keppra patient Submitted by NotafanofBran on Thu, 2008-12-11 - 03:22
LevETIRAcetam: Dosage, Mechanism/Onset of Action, Half-Life - Medicine.com Keppra: 100 mg/mL (473 mL) [gluten free, lactose free; contains methylparaben, propylparaben; grape flavor] Generic: 100 mg/mL (5 mL, 473 mL, 500 mL [DSC]) Tablet, Oral: Keppra: 250 mg [scored; contains fd&c blue #2 aluminum lake] Keppra: 500 mg [scored] Keppra: 750 mg [scored; contains fd&c yellow #6 aluminum lake] Keppra: 1000 mg [scored]
Off-label use of antiepileptic drugs for the treatment of neonatal ... Seventy-three percent (40/55) recommended treatment of neonatal seizures with one or both of levetiracetam and topiramate; 47% (26/55) recommended levetiracetam ...




















Komentar
Posting Komentar